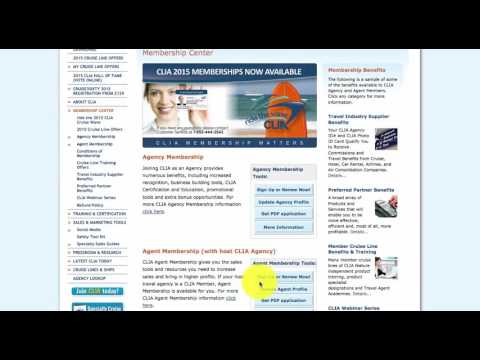
Hey team, this is Mike Rivera and I'm here to share with you how to get your CLIA card. Now, why would you need your CLIA card? One, it's great to have on you. It makes you look professional, and at the same time, it's gonna allow you to get lots of discounts from a lot of the vendors that we use. Now, if you're gonna go ahead and book a Disney Cruise as a travel agent or a Royal Caribbean cruise as a VIP cruise, then you definitely need your clear card in order to get that discount. So, how do you get a clear card? It's really simple. Go to cruising.org. Once you get here, click on "Agency and Agents." From here, you're going to go to the travel agent dashboard. You're going to enter in your CLIA number or personal ID. That number can be found in your back office under your travel section. The number is 45609642. You're gonna hit login. Once you're logged in, you're going to click on this banner "Clea 2015." From here, you're gonna sign up right here under agent membership. You're gonna enter your email information, your CLIA number again (456), and you're gonna hit submit continue. Now, it says here, "I hereby certify that I've earned five thousand or more in travel total cruise commissions." Don't worry about that. Extreme Travel will waive that so you can still apply for your CLIA card. Now, it's $49.95 or $9.99 a year for you to be a member, and they've got lots of benefits to be a part of CLIA membership. After a few days, you're gonna receive an email. That email is gonna say, "This is your 2015 CLIA membership guide." It's gonna have your personal ID and your personalized CLIA certificate, as well...
Award-winning PDF software





How to get a copy of my Clia Certificate Form: What You Should Know
What are FBI Requirements? You must meet the following requirements to receive a CIA certificate and access to the agency's collection of human biological materials: a. Knowledge of the collection, the purpose of collection, and the types of materials contained in the collection; b. Ability and motivation to comply with the requirements of FDA Standard Operating Procedures, regulations, and guidelines, including the Guidelines for the Responsible Custodian of Human Bioburden materials; c. Responsibility for determining the health risks and benefits, as appropriate, of the use of materials from this collection, including their potential used to support or promote the national security; d. Acceptable proficiency in the use of the information processing operations systems used by laboratory personnel who support the agency's acquisition of human biological materials; e. Appropriate knowledge and understanding of the principles of biosecurity and of the proper handling of materials from this collection when used in the employment of appropriate biosecurity procedures to protect the human health and well-being of the scientific community and the public; f. Ability and motivation to manage research, training, and professional activities consistent with the responsibilities assigned to the laboratory; g. Acceptable familiarity with the standards and procedures governing information security, including: (1) Procedures used to protect the confidentiality, integrity, and availability of agency information; (2) Requirements related to the safeguarding of information from unauthorized use, disclosure, or access; (3) Standards governing the management of personal identifying information (such as health information), and the appropriate handling of such information; (4) Standards and procedures to safeguard information used in developing and evaluating risk assessments, and the proper handling and storage of the results of these assessments; (5) Standards for the proper protection of the property rights of private persons or institutions; h. Acceptable familiarity and understanding of the provisions of the Federal Public Health Service Act, including: h-1. Requirements regarding the protection of human subject privacy pursuant to Section 1030(a)(2) of the Public Health Service Act; and h-3. Requirements related to the protection of human subject privacy and the appropriate use of personal identifiers of subjects of research. 3. CDC/CIA Certification and Agency-Specific Certification: A certified copy of the CDC certificate is valid for five years after the certificate is issued. A CDC-specific certificate is valid for two years and includes the following: i. Identification of the certificate holder; ii.
online solutions help you to manage your record administration along with raise the efficiency of the workflows. Stick to the fast guide to do Form CMS-116, steer clear of blunders along with furnish it in a timely manner:
How to complete any Form CMS-116 online: - On the site with all the document, click on Begin immediately along with complete for the editor.
- Use your indications to submit established track record areas.
- Add your own info and speak to data.
- Make sure that you enter correct details and numbers throughout suitable areas.
- Very carefully confirm the content of the form as well as grammar along with punctuational.
- Navigate to Support area when you have questions or perhaps handle our assistance team.
- Place an electronic digital unique in your Form CMS-116 by using Sign Device.
- After the form is fully gone, media Completed.
- Deliver the particular prepared document by way of electronic mail or facsimile, art print it out or perhaps reduce the gadget.
PDF editor permits you to help make changes to your Form CMS-116 from the internet connected gadget, personalize it based on your requirements, indicator this in electronic format and also disperse differently.
Video instructions and help with filling out and completing How to get a copy of my Clia Certificate